12/4/19 - Entrance Task Answer
|
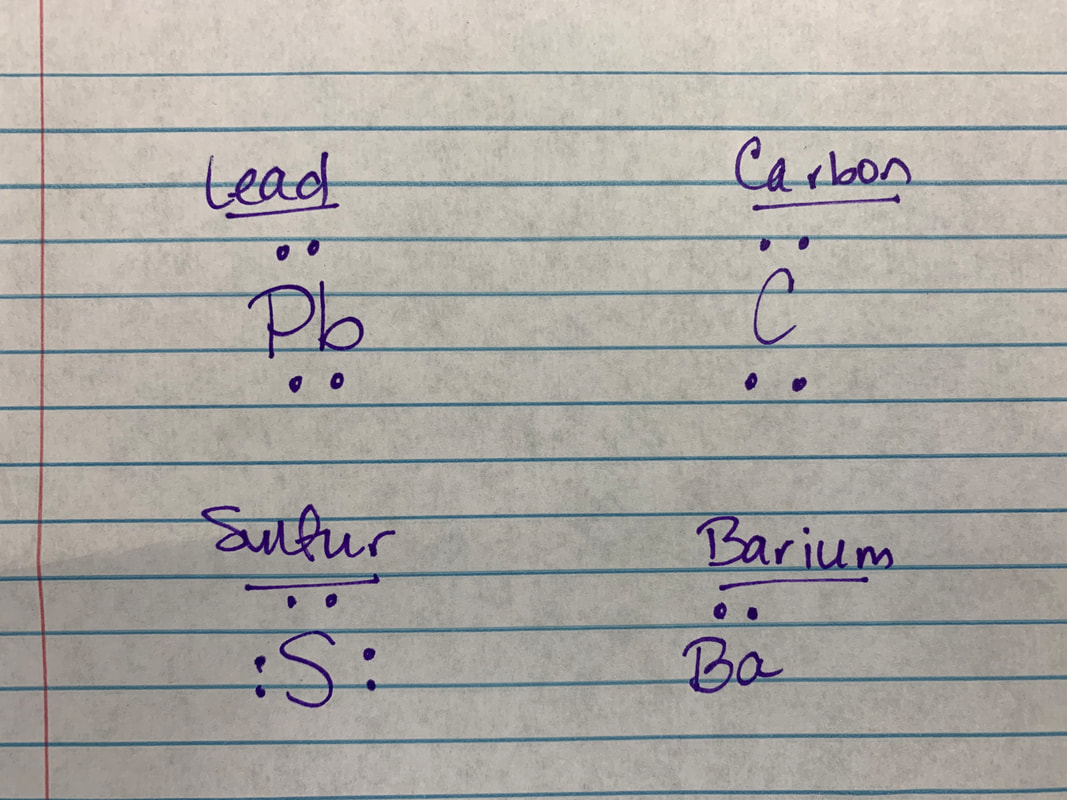
Ionic - Sulfur and Barium
Sulfur only needs 2 electrons to fill its outer shell and therefore has a stronger pull than Barium, which only has 2 valence electrons. Ionic bonds are formed between metals and nonmetals. The name of the substance would be Barium Sulfide. (remember - the metal comes first, the nonmetal second and changes the last part to -ide) Covalent - Lead and Carbon Lead and Carbon both have similar valence electrons and therefore similar needs to fill their outer shell. So they share electrons instead of giving away or taking. -------------------- |
Atoms have something called "electronegativity." Elements are more electronegative as you go further to the right and the further up the Periodic Table. So, something that is more electronegative (like Sulfur) is going to take electrons from something less electronegative (like Barium). Lead and Carbon have similar electronegativities, so they will share.
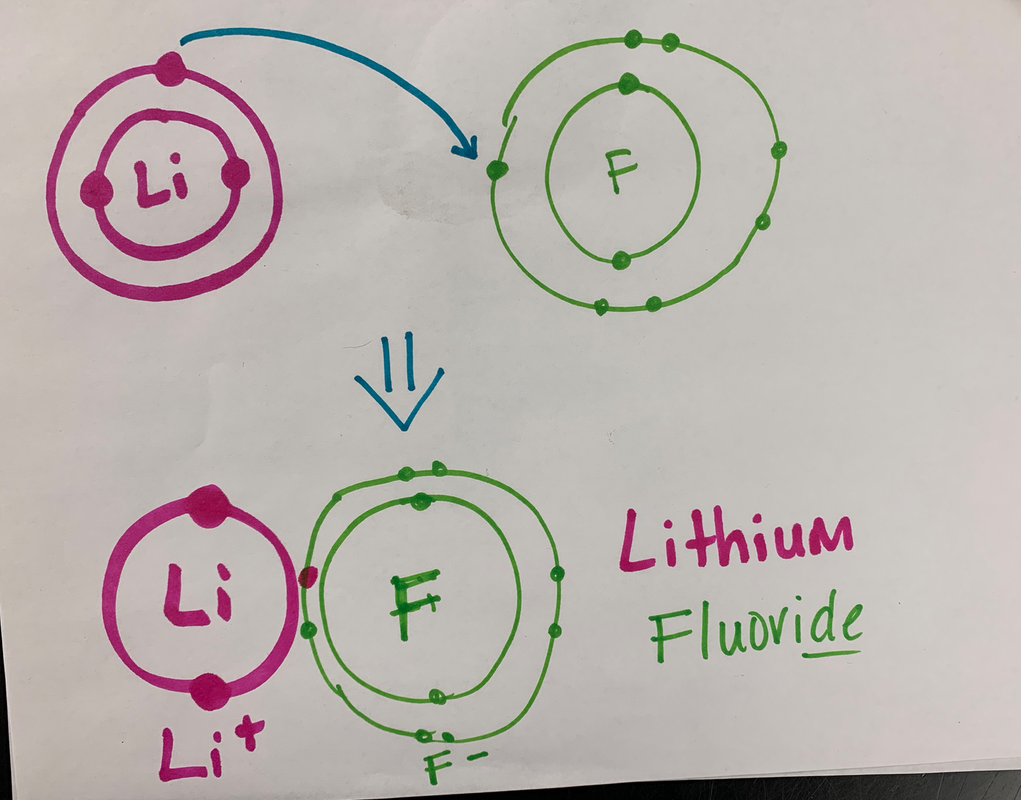
12/3/19 Entrance Task Answer
|
Answer: an Ionic Bond
Ionic bonds are formed between metals and nonmetals. In this case, Lithium is the metal and Fluorine is the non-metal. The Fluorine is stronger than the Lithium, and almost has a full shell, so it pulls the Lithium's electron away. The Lithium atom becomes the cation (positive ion) and is positive because it has lost an electron. The Fluorine atom becomes the anion (negative ion) and is negative because it has gained an electron. The name is this molecule is Lithium Fluoride. |
11/25/19 - Entrance Task Answer
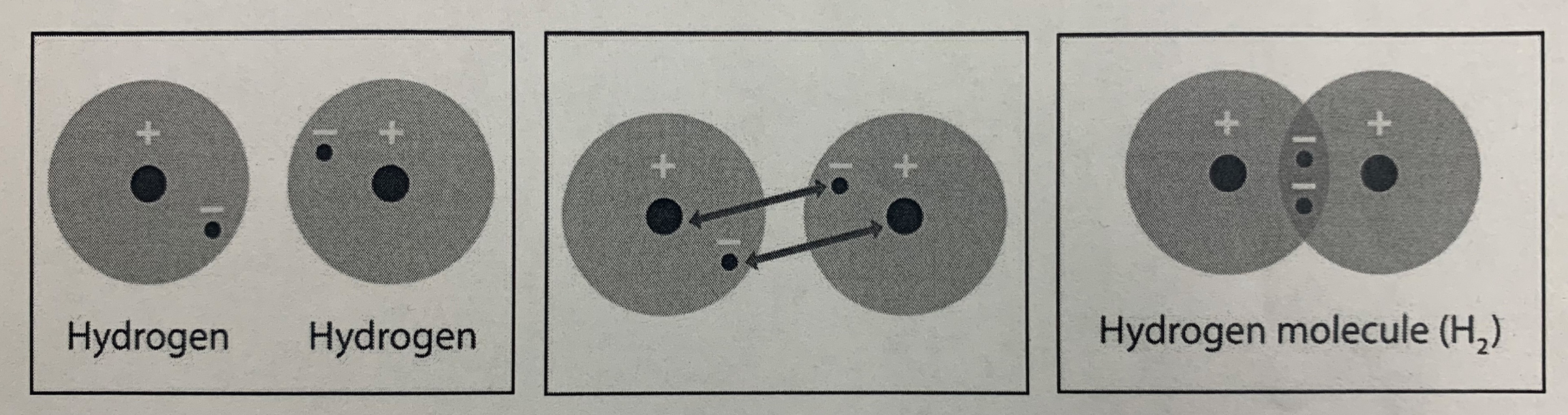
11/20/19 - Entrance Task Answer
|
Answer: Covalent Bond
Reasoning: It is a covalent bond because the electrons in each Hydrogen atom are being shared, not borrowed like an ionic bond. Hydrogen only has one electron shell, which holds 2 electrons. Remember - atoms want to be "happy" and have their outermost shell filled. So Hydrogen needs 2 electrons in order to be happy! |