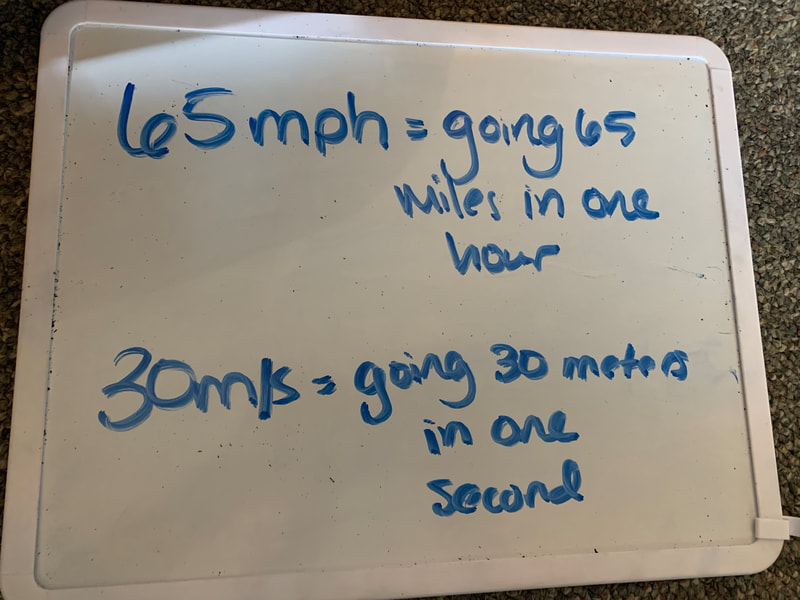April 27 Update on our Class Structure for the Remainder of the Year!
- I’m going to be doing demonstrations on Google Meet every Tuesday from 1-2! They will not last the entire hour, but students should try to log in right at 1 so we can start promptly! Each demo will have a short reflection due the following Friday. I will post a video of the demo on Classroom and our website if anyone is not able to make the Google Meet on Tuesday. I will send the link to our meeting to students in an email before our first meeting.
- Thursdays, I will do a help session on Google Hangouts from 1-2 to answer any questions students may have about any of the assignments. It is not required, but I will be able to answer questions immediately! I am also open to creating more help sessions in Google Hangouts if students can’t make this time frame.
- One of the most important - students should NOT spend more than 30-45 minutes per day on work for our class! My goal is to do short assignments with some longer projects built in. If students are unable to access something that has been posted, please email me immediately so we can get it figured out! :)
Monday, April 27 - Friday, May 1
Google Classroom - assignment posted on Monday of videos about speed, velocity, and momentum. Take notes from the video on our notes template (copy of it on Google Classroom). Notes, Keywords and Summary are due Friday, May 1
Tuesday - Demo on Velocity and Momentum via Google Meet from 1-2. (It will not take the entire hour). This week's demo does not have a reflection due to some unforeseen technical difficulties!
Speed and Momentum Lab - will be posted on Google Classroom on Tuesday and will be due the following Monday, May 4.
Below is a "How-to Guide" for the speed and momentum lab, as well as some information on how to calculate speed/velocity and momentum. These were the focus of our demo on Tuesday, April 28.
Tuesday - Demo on Velocity and Momentum via Google Meet from 1-2. (It will not take the entire hour). This week's demo does not have a reflection due to some unforeseen technical difficulties!
Speed and Momentum Lab - will be posted on Google Classroom on Tuesday and will be due the following Monday, May 4.
Below is a "How-to Guide" for the speed and momentum lab, as well as some information on how to calculate speed/velocity and momentum. These were the focus of our demo on Tuesday, April 28.
Monday, April 13-Thursday, April 16
Standards/Proficiencies
MS-PS2-1. Apply Newton’s Third Law to design a solution to a problem involving the motion of two colliding objects.*
MS-PS2-2. Plan an investigation to provide evidence that the change in an object’s motion depends on the sum of the forces on the object and the mass of the object.
MS-PS2-1. Apply Newton’s Third Law to design a solution to a problem involving the motion of two colliding objects.*
MS-PS2-2. Plan an investigation to provide evidence that the change in an object’s motion depends on the sum of the forces on the object and the mass of the object.
Schoolwork:
1. Tuesday or Thursday - Google Hangout Review Session from 1-2pm (will send out a link via email to all of my 8th graders!) - It will probably not last the entire time, but will start at 1pm!
Please email me with any questions or concerns!
1. Tuesday or Thursday - Google Hangout Review Session from 1-2pm (will send out a link via email to all of my 8th graders!) - It will probably not last the entire time, but will start at 1pm!
- We will use a program called "Nearpod" to deliver review information! Students will take notes in our typical notes format (there will be a digital copy on Google Classroom or students can create their own in a notebook or other piece of paper)
- Review Topics:
- Newton’s 1st, 2nd, and 3rd Laws Newton’s
- First Law - An object in motion stays in motion, and an object at rest stays at rest, unless an unbalanced force acts on it.
- Newton’s Second Law - F=ma… the acceleration of an object depends on the force upon the object as well as the object’s mass.
- Newton’s Third Law - For every action, there is an equal and opposite reaction.
- Homework (Due Thursday, April 16) - Do Keywords/Questions and 3-5 Sentence Summary from the notes/review session
- Newton’s 1st, 2nd, and 3rd Laws Newton’s
- Find the list of activities to try at home and CHOOSE ONLY 1. Then, reflect on the activity in the Google Doc on the assignment on how each of Newton's 3 laws apply to it. This assignment is due Thursday, April 16.
Please email me with any questions or concerns!
Google Classroom Assignment
March 23-27 - Newton's 1st and 2nd Laws
Standards/Proficiencies:
MS-PS2-1. Apply Newton’s Third Law to design a solution to a problem involving the motion of two colliding objects.*
MS-PS2-2. Plan an investigation to provide evidence that the change in an object’s motion depends on the sum of the forces on the object and the mass of the object.
MS-PS2-1. Apply Newton’s Third Law to design a solution to a problem involving the motion of two colliding objects.*
MS-PS2-2. Plan an investigation to provide evidence that the change in an object’s motion depends on the sum of the forces on the object and the mass of the object.
Assignments:
- Section 1 of the Forces and Motion Packet (Newton's 1st and 2nd Laws)
- Google Classroom Assignment - Force and Motion activities to try at home (due Monday, 3/30)
March 18-April 6 ClassWork
- Newton’s Laws and Forces and Motion Packet
- Read and answer questions on the three sections of reading about Newton’s Laws and Forces. There are also questions embedded in the reading to check for understanding. Make sure you are doing these also!
- There are some math questions that go with the reading. I provided some equations and some notes to help you or help you figure out how to solve it.
- I will be posting some other videos on Google Classroom to supplement these questions. Please let me know if you cannot find or access these videos!
- Read and answer questions on the three sections of reading about Newton’s Laws and Forces. There are also questions embedded in the reading to check for understanding. Make sure you are doing these also!
Newton's Laws!
Save the Penguins Powerpoint
1/30/20 - Thermal Energy Organizer
Click on the link below labeled "Thermal Energy." There are four main sections:
Then, under "Conclusion," write a 3-5 sentence summary of your notes. Don't just repeat your notes. Try to reword them.
Thermal Energy <------- Click here!!!
- In a Flash
- Burning Questions
- Examples of Thermal Energy
- What do you Mean?
Then, under "Conclusion," write a 3-5 sentence summary of your notes. Don't just repeat your notes. Try to reword them.
Thermal Energy <------- Click here!!!
1/27/20 - Reading and Questions while Ms. Saikin is out
Directions: You will read an article on Edji and answer several questions on Google Classroom that go along with the article. Highlight the evidence from the article that helps you answer the questions.
1. Read the article "Invest in the Nest" on Edji. Clic below to get to Edji
Edji - Code: c5fx
2. Answer the questions on the Google Doc in Google Classroom titled "Invest in the Nest Reading Questions" as you read through the article.
1. Read the article "Invest in the Nest" on Edji. Clic below to get to Edji
Edji - Code: c5fx
2. Answer the questions on the Google Doc in Google Classroom titled "Invest in the Nest Reading Questions" as you read through the article.
12/4/19 - Entrance Task
1. Title your journal page "Ionic or Covalent part 2?" Divide your page down the middle (like yesterday) and label the left side "My first thoughts" and the right side "My New Thoughts).
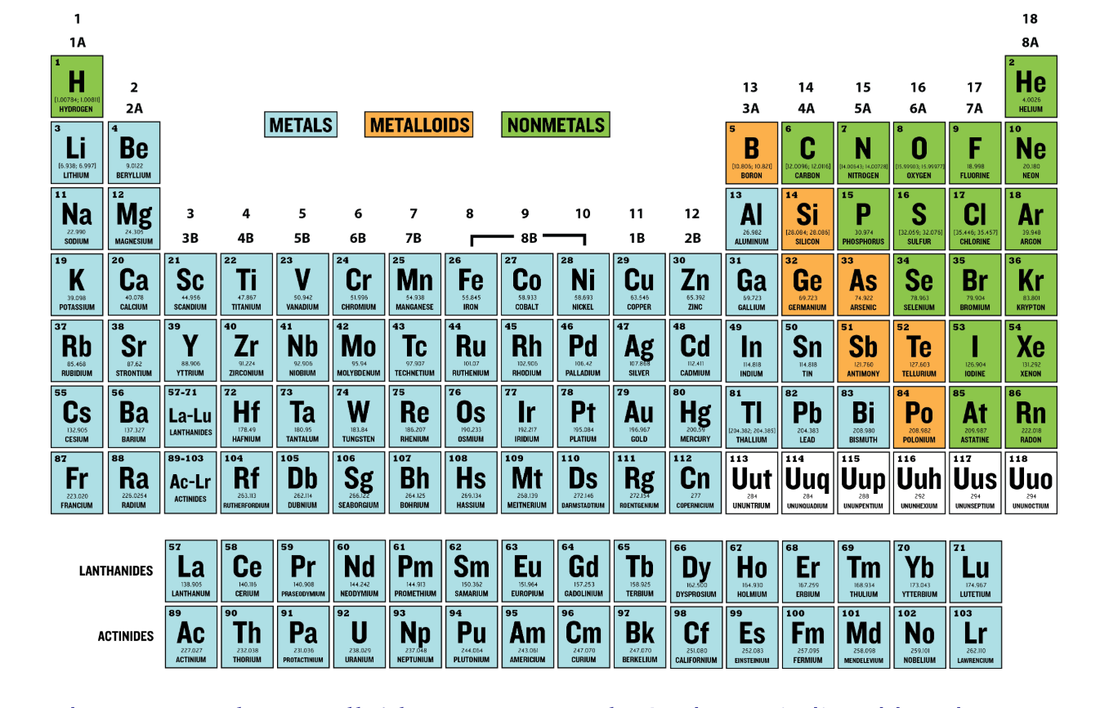
2. On the left side - Draw Lewis Dot Structures for the following elements. Indicate if they are metals, non-metals, or metalloids (you are going to write more, so don't fill up this whole side yet)
-Lead
-Sulfur
-Carbon
-Barium
3. Also on the left side - Which atoms do you think will form covalent bonds and which will form ionic? Why do you think that?
4. When you are done, click the link below to find the real answer.
Entrance Task Answer
2. On the left side - Draw Lewis Dot Structures for the following elements. Indicate if they are metals, non-metals, or metalloids (you are going to write more, so don't fill up this whole side yet)
-Lead
-Sulfur
-Carbon
-Barium
3. Also on the left side - Which atoms do you think will form covalent bonds and which will form ionic? Why do you think that?
4. When you are done, click the link below to find the real answer.
Entrance Task Answer
12/3/19 Entrance Task
1. Title your journal page "Ionic or Covalent?" Divide your page down the middle (like yesterday) and label the left side "My first thoughts" and the right side "My New Thoughts).
2. Answer on the left side. What type of bond is going to be formed in the picture below? Will it be ionic or covalent? Why do you think that? Think about what you know about the valence electrons. Draw the resulting bond between the atoms as part of your answer. What is the name of the molecule?
3. When you're done, check the answer below. Include any information you didn't have in the right side!
2. Answer on the left side. What type of bond is going to be formed in the picture below? Will it be ionic or covalent? Why do you think that? Think about what you know about the valence electrons. Draw the resulting bond between the atoms as part of your answer. What is the name of the molecule?
3. When you're done, check the answer below. Include any information you didn't have in the right side!
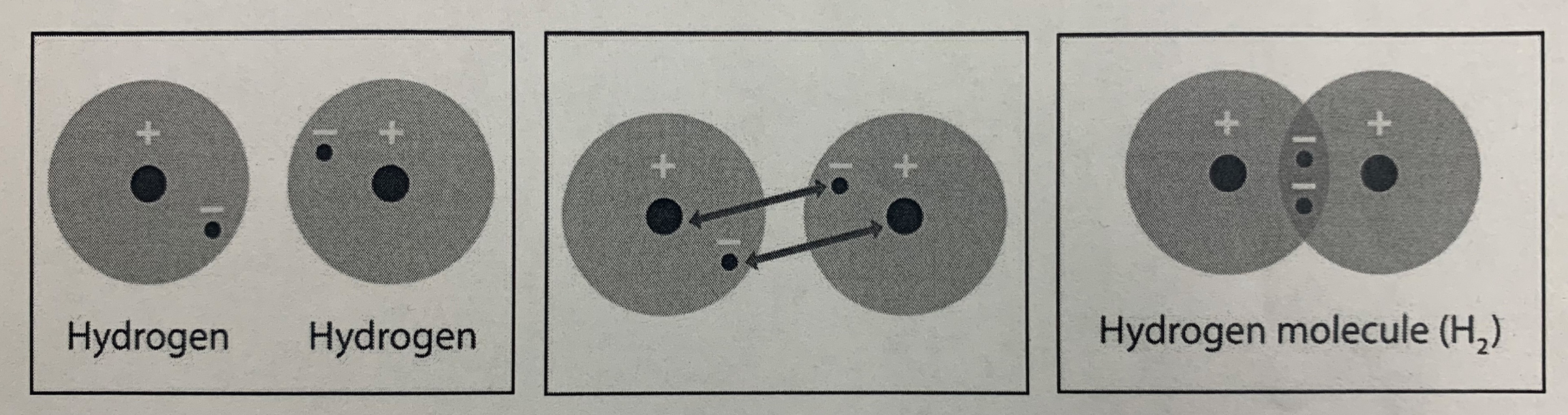
11/25/19 - Covalent Bonds and Lewis Dot Structures
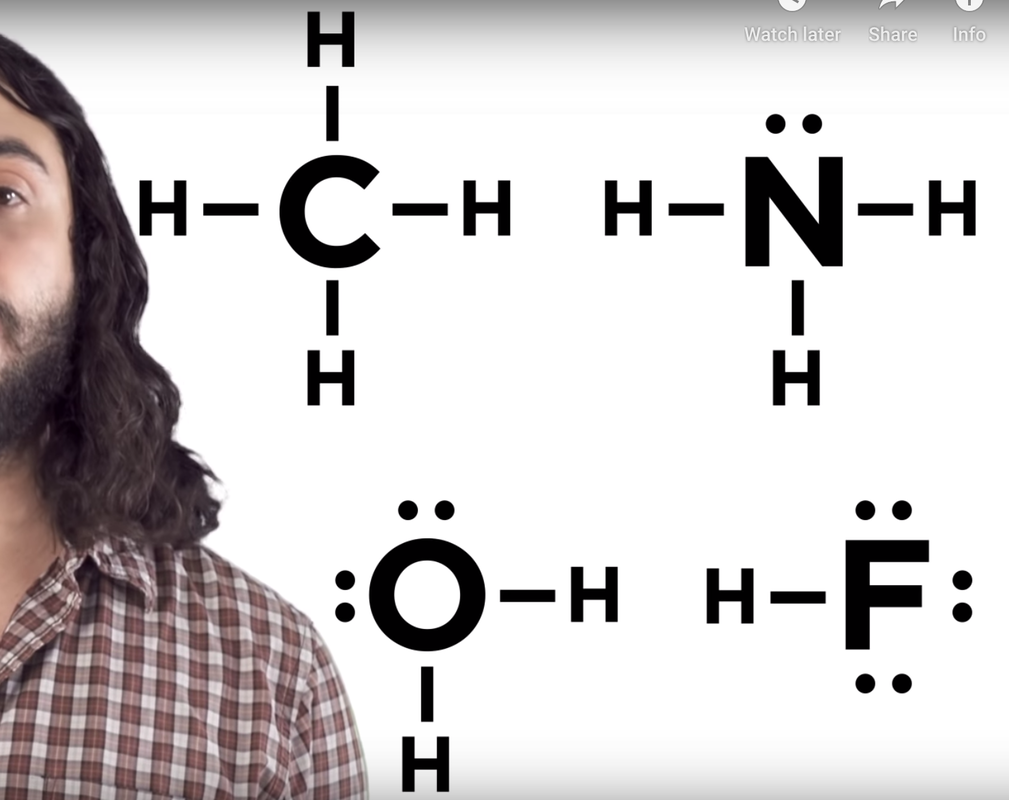
Title your page "Covalent Bond Lewis Dot Structures.
Write the four chemical formulae on the left in your journal. Then, using the second picture as reference, try draw lewis dot structures of those covalent bonds. Remember - the covalent bond represents 2 valence electrons. Think about how many valence electrons each atom has. Also remember the octet rule - atoms (except Hydrogen and Helium, which want 2). When you're done, check in with Ms. Saikin and then click the answer below.
Write the four chemical formulae on the left in your journal. Then, using the second picture as reference, try draw lewis dot structures of those covalent bonds. Remember - the covalent bond represents 2 valence electrons. Think about how many valence electrons each atom has. Also remember the octet rule - atoms (except Hydrogen and Helium, which want 2). When you're done, check in with Ms. Saikin and then click the answer below.
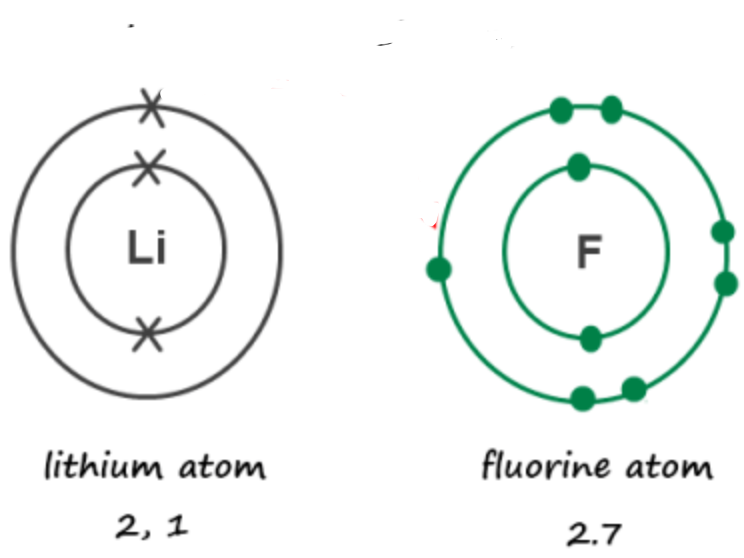
11/22/19 - Periodic Table for Ionic Bonding
Title your page "Ionic Bonding"
Grab a periodic table from the back table and glue/tape it into your journal.
Color the periodic table following the diagram below. You only need 3 colors!
...That's it! :)
Grab a periodic table from the back table and glue/tape it into your journal.
Color the periodic table following the diagram below. You only need 3 colors!
...That's it! :)
11/21/19 Entrance Task - Carbon Dioxide
Title your page "Carbon Dioxide Covalent Bond"
Recreate the drawing in your journal.
Underneath your drawing, explain why carbon dioxide has covalent bonds in terms of the valence electrons. How do the carbon and oxygen atoms become "happy?"
We will go over the answer as a class!
Recreate the drawing in your journal.
Underneath your drawing, explain why carbon dioxide has covalent bonds in terms of the valence electrons. How do the carbon and oxygen atoms become "happy?"
We will go over the answer as a class!
This video will be for our lesson today.
11/20/19 Entrance Task - What type of Bond?
1. Title your journal page "What type of bond?" Divide your page down the middle (like yesterday) and label the left side "My first thoughts" and the right side "My New Thoughts).
2. Recreate the drawing below in your journal on the left side.
3. Answer the following question (still on the left side) - Based on what you read yesterday on the edji article about bonding, explain what type of bond (ionic or covalent) you think this is and why. Think about what is happening with the electrons in order to create this molecule. (you may go back into your notes from yesterday or go back to the edji article to help you. Use your resources!)
4. When you are done, check in with Ms. Saikin. Then, you'll be able to check the correct answer by clicking the link below the picture. Put the correct information (whether you got it right or wrong) on the right side in your own words.
2. Recreate the drawing below in your journal on the left side.
3. Answer the following question (still on the left side) - Based on what you read yesterday on the edji article about bonding, explain what type of bond (ionic or covalent) you think this is and why. Think about what is happening with the electrons in order to create this molecule. (you may go back into your notes from yesterday or go back to the edji article to help you. Use your resources!)
4. When you are done, check in with Ms. Saikin. Then, you'll be able to check the correct answer by clicking the link below the picture. Put the correct information (whether you got it right or wrong) on the right side in your own words.
Here's the correct answer! Make sure you've checked with Ms. Saikin first!
11/19/19 - Bonding - Double Entry Journal
In your journal, open up to your next blank page. Title the page "Bonding".
Divide your page in half down the middle from top to bottom. Label the left side "My First Thoughts." Label the right side "My New Thoughts"
On the left side, take a few minutes to write what you think you know about bonding (why do atoms bond together to make molecules?). This will be hard. It's okay. Be creative with your answer. Just tell me (or draw!) what you think you know. Close your journal when you're done.
Read the article below. Highlight evidence to support your new explanation of why atoms bond together. Then, in the right column, write and/or draw why atoms bond together. You may work alongside someone at your table to do this (I encourage you)
https://edji.it/
Divide your page in half down the middle from top to bottom. Label the left side "My First Thoughts." Label the right side "My New Thoughts"
On the left side, take a few minutes to write what you think you know about bonding (why do atoms bond together to make molecules?). This will be hard. It's okay. Be creative with your answer. Just tell me (or draw!) what you think you know. Close your journal when you're done.
Read the article below. Highlight evidence to support your new explanation of why atoms bond together. Then, in the right column, write and/or draw why atoms bond together. You may work alongside someone at your table to do this (I encourage you)
https://edji.it/
11/18/19 - Plans for when Ms. Saikin is out
Click on the link below to watch a TED Ed video about plastic bottles as trash. There are questions for you to answer based on the video. If paper copies of these questions are not available, answer the questions in your journal. They are provided below! They must be answered in complete sentences. Some come right from the video, others you have use what you know or think you know to answer them.
Plastic Bottles
Plastic Bottle TED ED questions
1. Explain how plastic is made. Be as specific as you can, including the molecules that make them.
2. What do you think “monomers” and “polymers” are? Look at what they show you in the video.
3. Explain what happens in landfills when it rains. What happens to the water-soluble particles?
4. What does “water soluble” mean? Again, watch what’s happening in the video to help you with your answer.
5. What is “leachate"?
6. How long does it take plastic in landfills to decompose?
7. What are the gyres in the ocean? How are they created?
8. How is plastic recycled?
If you finish the questions, continue to update your journal with the cutouts we started on Friday!
Plastic Bottles
Plastic Bottle TED ED questions
1. Explain how plastic is made. Be as specific as you can, including the molecules that make them.
2. What do you think “monomers” and “polymers” are? Look at what they show you in the video.
3. Explain what happens in landfills when it rains. What happens to the water-soluble particles?
4. What does “water soluble” mean? Again, watch what’s happening in the video to help you with your answer.
5. What is “leachate"?
6. How long does it take plastic in landfills to decompose?
7. What are the gyres in the ocean? How are they created?
8. How is plastic recycled?
If you finish the questions, continue to update your journal with the cutouts we started on Friday!
10/25/19 - Chromatography Notes
10/21/19 - TED Talks while Ms. Saikin is gone
Choose 3 of the 5 TED Talks below and fill in the organizer to go with them.
|
What is the Universe Made of?
|
The Chemistry of Cookies
|
Just How Small is an Atom?
|
|
Is Fire a Solid, Liquid, or a Gas?
|
How Do We Smell?
|
10/17/19 - Build a Molecule
10/9/19 Build an Atom
9/26/19 - TED Talks while Ms. Saikin is out
Watch the three videos below and fill in the TED Talk Organizer based on each video.
|
|
|
|
9/25/19 - Periodic Table
Scientific Writing Practice - Commercials!
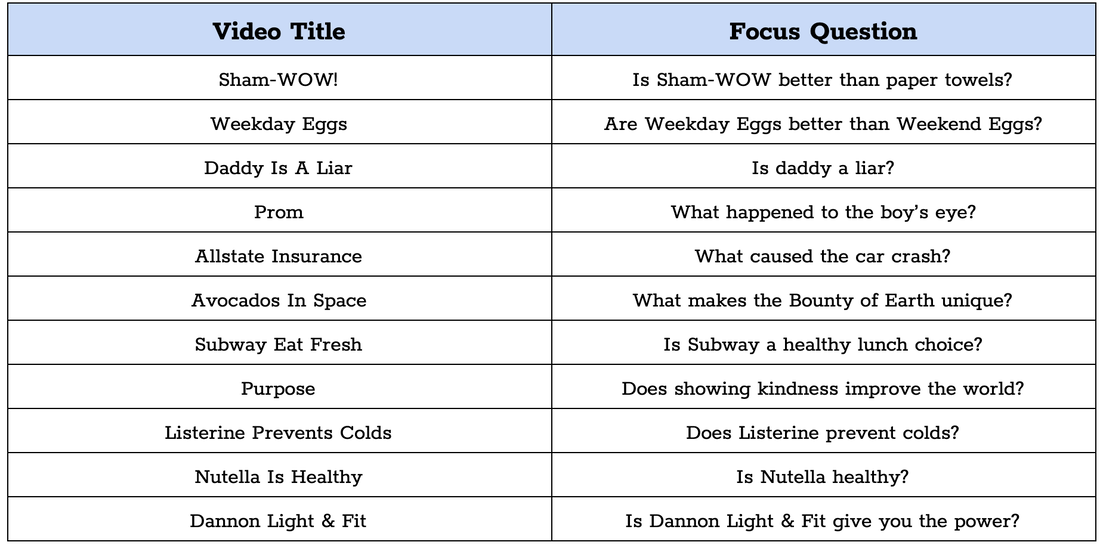
You are going to practice some scientific writing using commercials. We are going to write what's called a CER, which stands for Claim, Evidence, and Reasoning. We are going to do one together as a group, then you'll do some practice on your own!
Click on the link below to gain access to the commercials. You are going to watch two commercials and fill out the graphic organizer by answering the question below about the commercials you watch.
Padlet: Commercials for CER Practice
Padlet: Commercials for CER Practice